-
C-24 configuration in stigmasterol
Thursday, August 27, 2009 -
Determination of the configuration at C-24 in stigmasterols by comparison of 13C NMR spectra
Example: 3-Hydroxystigmast-5-en-7-one (CDCl3, 77.0 ppm)
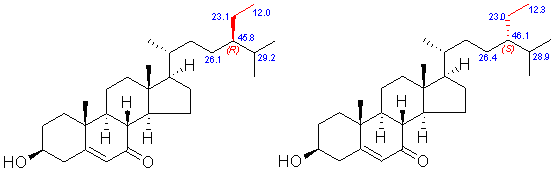
Without substitution or oxygenation on D-ring and side chain, this rule is highly reliable. It should be noted that the C-24 epimers are identical in TLC behavior.
Notes
-
JCR 2010 --- Natural Product Chemistry
Wednesday, June 29, 2011 -
neo-Clerodane diterpenoids from Scutellaria barbata
Friday, November 19, 2010 -
Bi-linderone and linderaspirone A
Wednesday, July 21, 2010 -
JCR 2009 --- Natural Product Chemistry
Tuesday, June 22, 2010 -
Iridoid glycosides from Callicarpa
Friday, April 9, 2010 -
ent-Pimaranes from Siegesbeckia orientalis
Saturday, December 5, 2009 -
Structure revision of an isoflavonoid
Sunday, November 1, 2009 -
C-24 configuration in stigmasterol
Thursday, August 27, 2009 -
JCR 2008 --- Natural Product Chemistry
Monday, June 22, 2009 -
A novel A-ring contracted oleanane triterpenoid
Sunday, May 31, 2009